COVID-19 SARS-CoV-2 Neutralizing Antibody Assay Kit
SARS-CoV-2 Neutralizing Antibody Assay Kit (Fluorescence Immunochromatography) is used to quantitatively detect the 2019-nCoV neutralization antibodies in clinical samples (serum asmal/whole blood).
What are COVID-19 neutralizing antibody rapid tests?
● COVID-19 SARS-CoV-2 Neutralizing Antibody Assay Kit is a point of care test for use in multiple testing locations and has high sensitivity and specificity. Meaning it has the potential to play a vital supporting role out of the pandemic.
● Screen for an immune response following COVID-19 infection or vaccination
● A testing solution for multiple scenarios: workplace testing, border control or vaccine rollout
● Results in 15 minutes from a small blood sample
What are neutralizing antibodies?
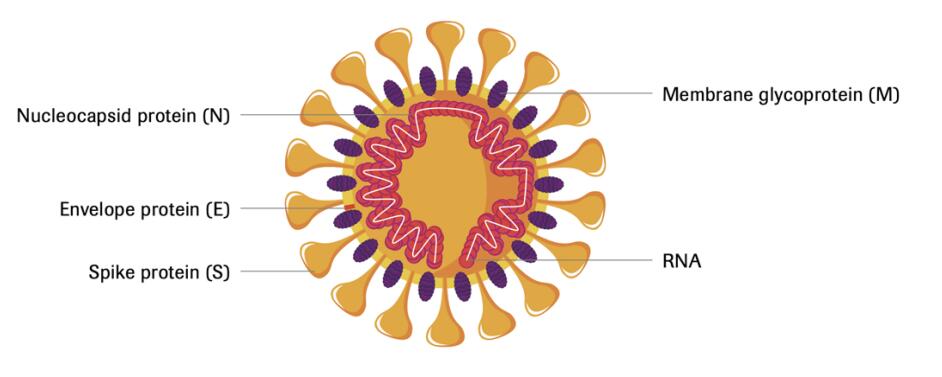
Following infection, the body produces antibodies targeting specific parts of the SARS-CoV-2 (COVID-19) virus with the Nucleocapsid protein and the Spike protein being of importance in the context of antibody tests. The coronavirus nucleocapsid protein is a multi-functional protein1 and the spike protein is integral in penetrating human cells and initiating infection.
References
1.McBride, R et al.The Coronavirus Nucleocapsid Is a Multifunctional Protein. Viruses. 2014 Aug; 6(8): 2991- -3018. doi: 10.3390/v6082991.
Neutralizing Antibody Rapid Test Intended Use
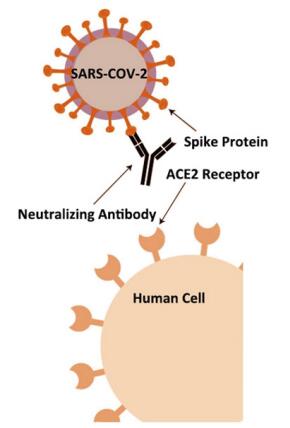
Neutralizing Antibody Rapid Test Intended Use spike (S), envelope (E), membrane (M), and nucleocapsid (N). The S-protein contains a receptor binding domain (RBD), which can recognize the cell surface receptor, angioten- sin-converting enzyme-2 (ACE2). In a recent study, neutralizing antibody (NAb) can block the interaction between the receptor-binding domain (RBD) of the novel coronavirus spike protein with the ACE2 cell surface receptor. The level of NAb, therefore, can be used to analyze a patient's immunity against future SARS-CoV-2 infection. This COVID-19 neutralizing antibody lateral flow assay rapidly detects any antibodies that can neutralize the RBD-ACE2 interaction.
Structure of the Severe Acute Respiratory Syndrome
Coronavirus 2(SARS-Cov-2)
Operation Steps

- +852-3500-5196
- info@kindmay.com
-
32nd Floor, Block B, Building 10,
Shenzhen Bay Science & Technology Ecological Park,
Nanshan, Shenzhen 518057 China

 Language
Language



